July 10, 2023 • 4 min read
Optimizing Clinical Trials with GS1’s New Application Standard
CATEGORIES
SOCIAL SHARE
Introduction to GS1’s Latest Innovation
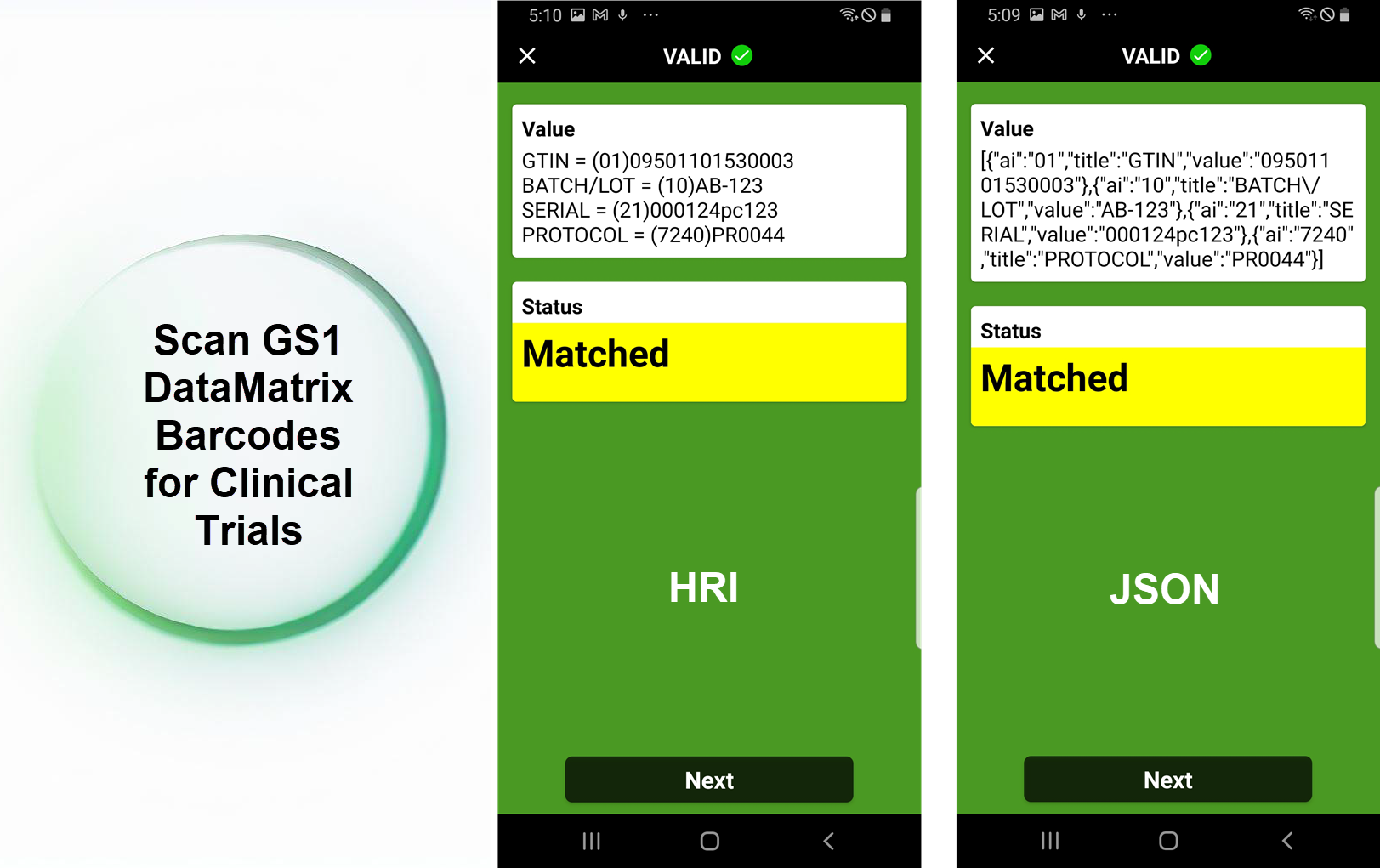
GS1 has launched the Application Standard aimed at revolutionizing the management and safety of investigational products in clinical trials. This new standard, developed through the collaboration of 60 experts from 37 clinical trial organizations globally, focuses on the identification and barcoding of these products. By facilitating seamless integration with edge computing technologies with CodeREADr, it promises to enhance the safety and efficiency of clinical research processes.
The Role of Edge Computing in Clinical Trials
Edge computing plays a critical role in clinical trials by processing data closer to its source, thus enabling faster and more effective decision-making. This technology involves a range of networks and devices that operate at or near the user, optimizing the handling of data in real time. In the realm of clinical trials, edge computing can significantly boost subject safety through real-time, actionable insights provided directly to clinicians.
Enhancing Subject Safety with Structured Data
The new GS1 Application Standard offers multiple safety benefits by utilizing structured data:
- Enrollment Validation: Clinicians can quickly verify a research subject’s identity by scanning their barcode ID, ensuring that only authorized individuals participate.
- Duplicate Dose Warning: Immediate alerts prevent administering multiple doses to a subject within a short interval, enhancing medication safety.
- Drug Verification: Accurate scanning of GS1 Data Matrix barcodes ensures that the medication administered matches the trial’s prescribed drugs.
- Dosage Verification: Discrepancies between the administered and prescribed dosages are instantly flagged, minimizing errors in drug delivery.
Future Prospects: Efficiency and Safety
The integration of GS1’s new standard with edge computing not only streamlines clinical trial processes but also leads to safer and more precise treatment administration. This forward-thinking approach is expected to expedite the delivery of new therapies and enhance patient outcomes. Looking ahead, we will explore additional advantages such as improved traceability and inventory management, which extend from the production of investigational products to their distribution and use in clinical settings.
The collaboration between GS1 and various clinical trial stakeholders marks a significant advancement in clinical trial management. By adopting this new standard, the industry can achieve higher safety standards and operational efficiencies, ultimately benefiting patients worldwide.


